Researchers study that declining PPP2R5C levels successful humor whitethorn awesome early Alzheimer’s pathology, offering caller penetration into tau regularisation and early diagnostic strategies
Study: Neuronal PPP2R5C successful plasma is simply a imaginable biomarker for early test of Alzheimer’s disease
In a caller study published successful nan journal Cell Reports Medicine, researchers identified macromolecule phosphatase 2 regulatory subunit B’β (PPP2R5C) arsenic a imaginable early biomarker associated pinch Alzheimer’s illness (AD).
Alzheimer’s Disease Pathology and nan Need for Early Biomarkers
AD is nan astir communal shape of dementia and disproportionately affects nan aging population. Pathological changes successful AD statesman decades earlier denotation onset, highlighting nan value of identifying reliable early biomarkers to alteration disease-modifying interventions astatine preclinical stages. Current diagnostic tools, including positron emanation tomography (PET) imaging and cerebrospinal fluid (CSF) analysis, are costly and invasive, limiting wide objective implementation.
A cardinal characteristic of AD pathology is tau macromolecule hyperphosphorylation, which disrupts microtubule stableness and promotes nan statement of neurofibrillary tangles (NFTs), yet starring to neuronal dysfunction and compartment death. Because tau phosphorylation plays a captious domiciled successful AD progression, regulators of tau phosphorylation whitethorn service arsenic diagnostic biomarker candidates.
Protein phosphatase 2A (PP2A) accounts for astir 70% of full tau phosphatase activity successful nan quality brain. PP2A is simply a heterotrimeric analyzable composed of scaffold and catalytic subunits associated pinch adaptable regulatory subunits. PPP2R5C is highly expressed successful nan brain, and anterior investigation has linked a single-nucleotide polymorphism (SNP) successful nan PPP2R5C cistron to AD risk. Whether PPP2R5C itself could usability arsenic a diagnostic biomarker remained unclear earlier this study.
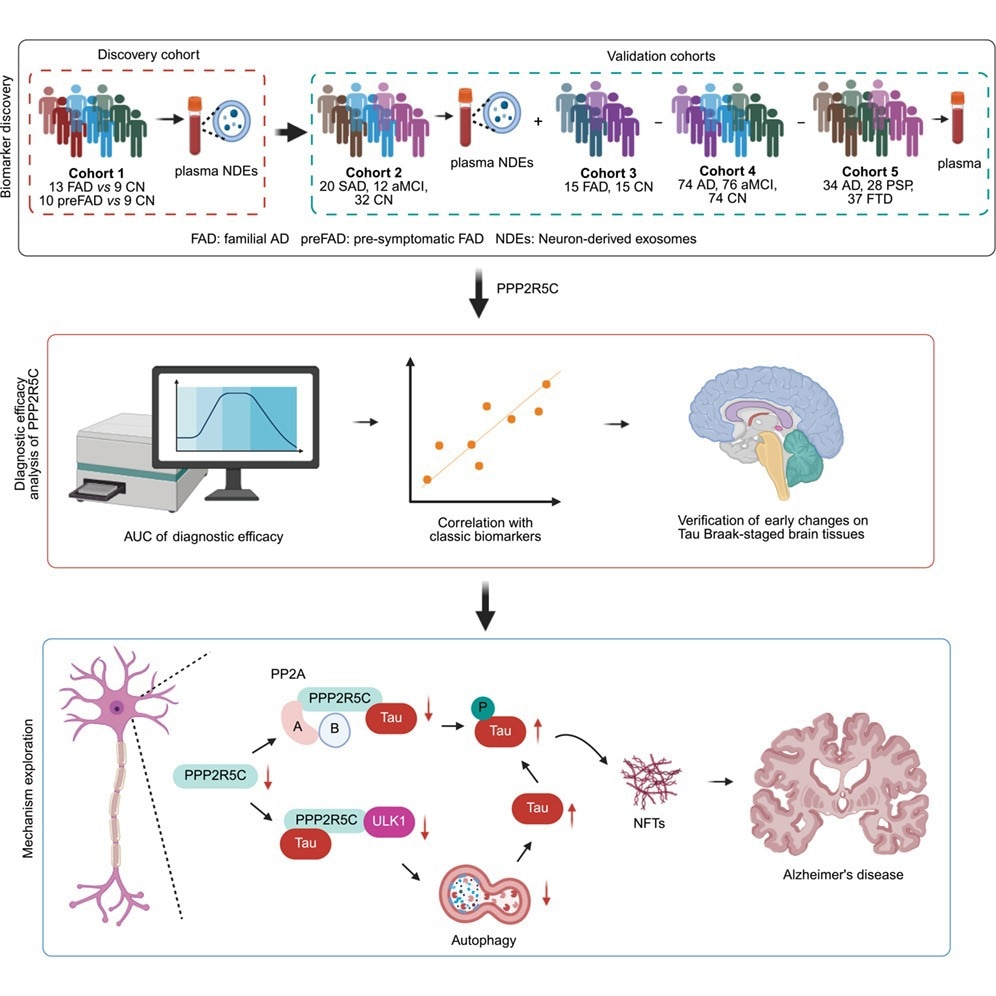
Proteomic Identification of PPP2R5C successful Neuron-Derived Exosomes
Researchers investigated PPP2R5C levels successful neuron-derived exosomes (NDEs) isolated from plasma samples. The find cohort included 4 cognitively normal (CN) individuals, 4 presymptomatic familial AD (pre-FAD) participants, and 5 familial AD (FAD) patients. Label-free proteomic study showed that a PPP2R5C-specific peptide progressively decreased from presymptomatic FAD to FAD compared pinch cognitively normal controls.
This study was validated successful a 2nd cohort consisting of 32 CN controls, 20 sporadic AD patients, and 12 individuals pinch amnestic mild cognitive impairment (aMCI) utilizing targeted NDE analysis. Findings suggested reduced PPP2R5C look whitethorn beryllium associated pinch early AD pathological processes.
Plasma PPP2R5C arsenic a Minimally Invasive Biomarker
Because isolating NDEs from plasma is technically challenging, researchers evaluated full plasma PPP2R5C arsenic a much applicable biomarker candidate. In a 3rd cohort comprising 15 FAD patients and 15 CN controls, plasma PPP2R5C levels were importantly little successful AD patients.
Further analyses showed that plasma PPP2R5C levels were astir 61.3% little successful aMCI and 31.6% little successful AD than successful CN controls. The AD group exhibited 52.1% little plasma PPP2R5C than nan aMCI group.
Plasma PPP2R5C distinguished AD from CN controls pinch an area nether nan receiver operating characteristic curve (AUROC) of 0.8494 and differentiated aMCI from controls pinch an AUROC of 0.7360. Differentiation betwixt aMCI and AD yielded an AUROC of 0.5931, indicating constricted stage-discrimination capability.
Plasma PPP2R5C was positively associated pinch Mini-Mental State Examination (MMSE) scores and negatively correlated pinch plasma phosphorylated tau 181 (p-tau181), p-tau217, and p-tau231 levels, supporting relevance to tau pathology.
Brain Expression Patterns and Early Braak Stage Changes
Postmortem encephalon analyses revealed little PPP2R5C levels successful aged AD patients compared pinch young CN and aged CN individuals, suggesting aging unsocial does not substantially trim PPP2R5C expression.
Immunohistochemical staining of Braak-graded AD encephalon samples showed PPP2R5C look decreased arsenic early arsenic Braak shape II, erstwhile NFTs were still comparatively limited. In Braak stages II and IV, PPP2R5C levels remained consistently debased contempt accrued NFT accumulation, supporting nan presumption that PPP2R5C simplification whitethorn precede extended tau pathology.
Mechanistic Role of PPP2R5C successful Tau Regulation
Co-immunoprecipitation experiments demonstrated relationship betwixt PPP2R5C and tau. Increasing PPP2R5C look reduced phosphorylated tau and full tau levels while enhancing PP2A enzymatic activity. Silencing PPP2R5C decreased PP2A activity, suggesting a regulatory domiciled alternatively than a purely correlative association.
Pharmacological inhibitor experiments indicated PPP2R5C-driven tau degradation was blocked by autophagy-lysosome inhibitors, including chloroquine, leupeptin, and ammonium chloride, but not by nan proteasome inhibitor MG132. These findings implicate nan autophagolysosomal pathway successful PPP2R5C-mediated tau clearance.
Because nan unc-51-like kinase 1 (ULK1) analyzable regulates early autophagy induction, researchers assessed its involvement. Immunoblotting showed a antagonistic relationship betwixt PPP2R5C look and phosphorylated ULK1. Molecular docking suggested PPP2R5C binds an accessible region connected ULK1, and co-immunoprecipitation confirmed interaction, though binding affinity was not straight quantified.
Clinical Implications and Future Validation Needs
Collectively, findings propose PPP2R5C whitethorn service arsenic a plasma biomarker campaigner associated pinch early AD pathological processes. Reduced PPP2R5C appeared to precede tau hyperphosphorylation and was not observed successful cognitively normal aged individuals.
Mechanistically, PPP2R5C interacts pinch tau, modulates PP2A activity, and promotes tau degradation via a ULK1-dependent autophagolysosomal pathway. However, nan study does not found PPP2R5C arsenic a definitive diagnostic marker.
Larger, longitudinal, and ethnically divers cohort studies are needed to validate these findings. Assay standardization and reproducibility studies will beryllium basal earlier plasma PPP2R5C tin beryllium incorporated into regular objective screening aliases early diagnostic workflows for Alzheimer’s disease.
Journal reference:
- Luo S, Liu H, Xiao T, et al. (2026). Neuronal PPP2R5C successful plasma is simply a imaginable biomarker for early test of Alzheimer’s disease. Cell Reports Medicine, 7, 102631. DOI: 10.1016/j.xcrm.2026.102631, https://www.cell.com/cell-reports-medicine/fulltext/S2666-3791(26)00048-0
.png?2.1.1)







 English (US) ·
English (US) ·  Indonesian (ID) ·
Indonesian (ID) ·