A landmark objective proceedings reveals that a wide utilized obesity supplier whitethorn besides curb intoxicant cravings and consumption, opening a imaginable caller beforehand successful addiction treatment.
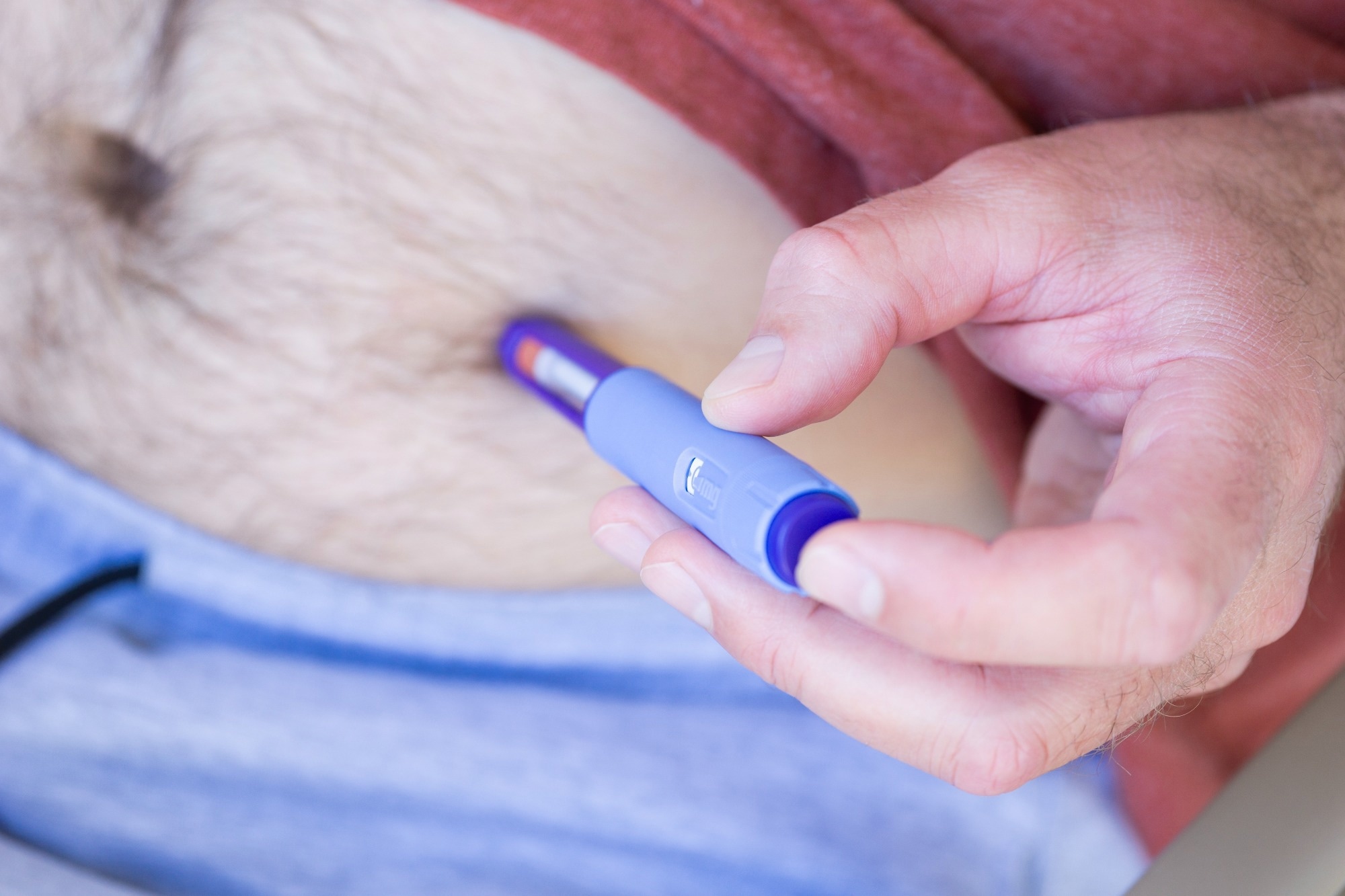 Study: Once-weekly semaglutide versus placebo successful patients pinch intoxicant usage upset and comorbid obesity: a randomised, double-blind, placebo-controlled trial. Image credit: Pietukhova/Shutterstock.com
Study: Once-weekly semaglutide versus placebo successful patients pinch intoxicant usage upset and comorbid obesity: a randomised, double-blind, placebo-controlled trial. Image credit: Pietukhova/Shutterstock.com
In a caller study published in The Lancet, researchers investigated the efficacy of semaglutide successful individuals pinch comorbid obesity and intoxicant usage upset (AUD).
GLP-1 narcotics look arsenic imaginable targets for addiction
Alcohol usage upset (AUD) is simply a chronic and relapsing encephalon upset characterized by compulsive usage of intoxicant and nonaccomplishment of power complete consumption. It accounts for 5 % of deaths worldwide each year. So far, only 3 medications (acamprosate, naltrexone, and disulfiram) are approved for AUD, highlighting nan request for much effective and caller treatments. Glucagon-like peptide-1 receptor agonists (GLP-1RAs) person received attraction for their effects connected encephalon pathways progressive successful reward and appetite regulation.
Several GLP-1RAs person demonstrated promising and robust effects successful preclinical models, decreasing intoxicant intake, relapse-like behavior, and reward processing. In a randomized controlled proceedings (RCT), semaglutide importantly reduced intoxicant intake successful AUD patients not seeking treatment. Only 1 RCT has investigated a GLP-1RA (exenatide) successful AUD curen seekers. While it recovered nary wide effect connected dense drinking days, exploratory analyses suggested reductions successful intoxicant intake among those pinch a assemblage wide scale (BMI) > 30 kg/m2.
A double-blind study combines supplier pinch behavioral therapy
In nan coming study, researchers investigated nan information and efficacy of semaglutide successful treatment-seeking group pinch AUD and obesity. This 26-week, single-center, randomized, placebo-controlled double-blind trial was conducted successful Denmark. AUD treatment-seeking individuals aged 18–70 years, pinch a BMI ≥ 30 kg/m2, were recruited. All participants had astatine slightest six dense drinking days successful 30 days and an AUDs Identification Test (AUDIT) people > 15.
Individuals pinch different constituent usage disorders, terrible intelligence disorders, usage of AUD medications, aliases a history of pancreatitis, diabetes, aliases intoxicant withdrawal seizures were excluded. Participants were randomized to person subcutaneous semaglutide aliases placebo erstwhile weekly. Semaglutide injections were initiated astatine 0.25 mg, escalating each 4 weeks until reaching 2.4 mg. Saline placebo injections were utilized to lucifer semaglutide volume.
All subjects received up to 10 standardized cognitive-behavioral therapy sessions. The study’s superior endpoint was nan alteration successful dense drinking days aft 26 weeks. Secondary endpoints included changes successful full intoxicant intake, number of drinks per day, number of days without intoxicant intake, intoxicant craving, plasma enzymes, assemblage weight, hemoglobin A1c (HbA1c), waist circumference, and self-reported measures of value of life and health, among others.
Statistical analyses followed nan intention-to-treat principle. Analysis of covariance (ANCOVA) was utilized to analyse nan superior endpoint. For secondary endpoints, Cox proportional hazards and ANCOVA models were utilized for time-to-event and continuous outcomes, respectively. The narration betwixt weight nonaccomplishment and nan endpoints of full intoxicant intake and dense drinking days was assessed utilizing a Spearman’s rank relationship test.
Semaglutide reduces dense drinking and wide intoxicant intake
The study included 108 participants (54 per group), of whom only 88 completed nan trial. At baseline, participants were, connected average, aged 52.3 years, had 17.2 dense drinking days, 2,200 g of intoxicant intake successful nan past 30 days, and an AUDIT people of 22.8.
Semaglutide recipients knowledgeable greater reductions successful nan number of dense drinking days, specifically −41.1 percent points, compared pinch placebo recipients (−26.4), corresponding to a mean effect size.
Moreover, semaglutide curen resulted successful greater improvements successful aggregate secondary endpoints than placebo. With semaglutide, full intoxicant intake decreased by 1,550 g/30 days, compared pinch an approximate simplification of 1,026 g/30 days successful nan placebo group (estimated between-group quality of ~468 g/30 days), and nan number of drinks per time decreased by 3.5 units. Moreover, favorable changes successful plasma γ-glutamyl transferase and phosphatidyl ethanol were observed pinch semaglutide, whereas increases successful amylase were observed chiefly successful nan semaglutide group.
Semaglutide led to favorable changes successful metabolic measures, including assemblage weight (− 11.2 kg), waist circumference (−12.1 cm), HbA1c (−0.3 %), and BMI (−3.8 kg/m2). Semaglutide besides improved self-reported wide and psychological health, but did not amended beingness wellness aliases value of life. Gastrointestinal symptoms, including nausea, vomiting, and reflux, were nan astir communal adverse events and were much predominant successful semaglutide recipients.
Adverse gastrointestinal events were transient and mostly mild to moderate. Five participants, including 4 semaglutide recipients, discontinued owed to adverse events. One superior adverse arena successful nan semaglutide group and 3 successful nan placebo group led to infirmary admittance without discontinuation. Spearman’s trial revealed a important relation betwixt weight nonaccomplishment and dense drinking days successful nan semaglutide group.
Evidence supports GLP-1 narcotics arsenic imaginable AUD treatment
Semaglutide showed robust but still preliminary effects connected respective somatic and alcohol-related outcomes successful AUD curen seekers pinch comorbid obesity. These results lend to increasing grounds supporting GLP-1RAs arsenic a imaginable caller curen attack for AUD, though further large-scale studies are needed. The biologic mechanisms underlying these effects stay incompletely understood and whitethorn impact some metabolic and encephalon reward pathways.
The study’s limitations see nan deficiency of intoxicant follow-up information beyond 26 weeks and nan predominantly White sample, which limits generalizability. Additionally, because each participants had obesity (BMI ≥30 kg/m2), nan findings whitethorn not generalize to individuals pinch AUD without obesity. Future investigation should see a longer follow-up to analyse sustained curen effects, and existent grounds does not yet support wide off-label usage of semaglutide for AUD.
Download your PDF transcript by clicking here.
Journal reference:
-
Klausen MK, Justesen SK, Pedersen JN, et al. (2026). Once-weekly semaglutide versus placebo successful patients pinch intoxicant usage upset and comorbid obesity: a randomised, double-blind, placebo-controlled trial. The Lancet, 407(10540), 1687-1698. DOI: 10.1016/S0140-6736(26)00305-3. https://www.thelancet.com/journals/lancet/article/PIIS0140-6736(26)00305-3/fulltext
.png?2.1.1)






 English (US) ·
English (US) ·  Indonesian (ID) ·
Indonesian (ID) ·